Dibutyltin(IV) formulations: Synthetic aspect, spectroscopic interpretation and computational calculation Scientific paper
Main Article Content
Abstract
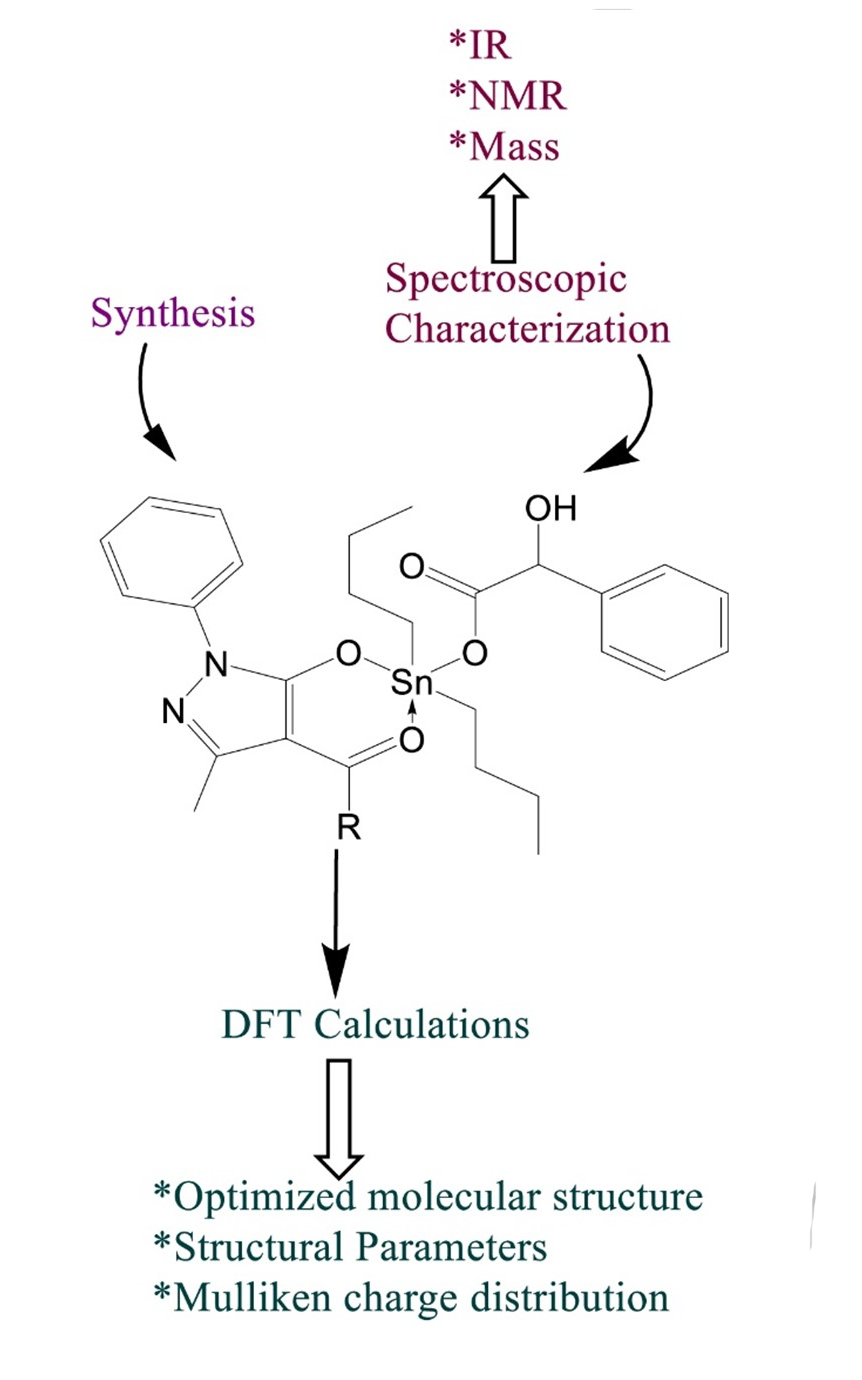
This research article reports a series of dibutyltin(IV) complexes, Bu2SnL(n)A (1a–d), synthesized by the reaction of sodium salts of substituted pyrazolones (L(n)H) and aromatic carboxylic acid (AH) with dibutyltin dichloride in dried THF solvent. The newly synthesized dibutyltin(IV) complexes were characterized by FT-IR, NMR (1H, 13C, 119Sn) spectroscopy and mass study. The coordination mode of both ligands with central tin atom was explicated with spectroscopic analysis of the complexes. The spectroscopic results revealed that the substituted pyrazolone ligand coordinates to the tin atom in a bidentate fashion via enolic oxygen and a carbonyl oxygen atom. In contrast, the aromatic carboxylic acid ligand coordinates in a monodentate manner through the carboxylic oxygen atom, resulting in a penta-coordinated environment around the central tin atom. Computational calculations using density functional theory (DFT) provide insights into the optimized molecular structures, energies, Mulliken charges and distortion in bond lengths and bond angles of the newly generated complexes. The global reactivity descriptors and the frontier molecular orbitals (FMOs) were calculated to understand the structural and electronic properties of dibutyltin(IV) formulations. The theoretical study explores the proposed penta-coordinated environment around tin atoms, revealing a distortion in the geometry of the complexes.
Downloads
Metrics
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.

Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution license 4.0 that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
References
K. Ullah, S. Ali, A. Haider, S. Naz, S. Yousuf, K.S. Munawar, M. S. Jan, R. Zafar, R. Kumar, J. Mol. Struct. 1322 (2025) 140444 (https://doi.org/10.1016/j.molstruc.2024.140444)
M. S. Mansour, A. T. Ibrahium, A.A. El‐Sherif, W. H. Mahmoud, Appl. Organomet. Chem. 38 (2024) e7656 (https://doi.org/10.1002/aoc.7656)
S. Budania, S. Saxena, A. Jain, J. Indian Chem. Soc. 101 (2024) 101198 (https://doi.org/10.1016/j.jics.2024.101198)
J. Devi, A. Boora, M. Rani, T. Arora, Anti-Cancer Agents Med. Chem. 23 (2023) 164 (https://doi.org/10.2174/1871520622666220520095549)
X. M. Du, J. W. Ma, Q. L. Li, Y. Cui, C. L. Ma, Appl. Organomet. Chem. 38 (2024) e7522 (https://doi.org/10.1002/aoc.7522)
D. A. Berseneva, D. B. Shpakovsky, E. A. Nikitin, V. E. Goncharenko, Y. A. Gracheva, K. A. Lyssenko, Y. F. Oprunenko, E. R. Milaeva, Russ. J. Coord. Chem. 49 (2023) 622 (https://doi.org/10.1134/S1070328423600559)
S. Rahim, A. Sadiq, A. Javed, M. Kubicki, B. Kariuki, M. Assad, N. Muhammad, N. Fatima, M. Khan, A. F. AlAsmari, F. Alasmari, J. Mol. Struct. 1313 (2024) 138703 (https://doi.org/10.1016/j.molstruc.2024.138703)
M. Pooyan, Z. Shariatinia, F. Mohammadpanah, K. Gholivand, M. Dusek, V. Eigner, M. Satari, A. A. E. Valmoozi, J. Mol. Liq. 391 (2023) 123442 (https://doi.org/10.1016/j.molliq.2023.123442)
S. Ramzan, S. Rahim, S. T. Hussain, K. B. Holt, J. K. Cockcroft, N. Muhammad, Z. Ur-
-Rehman, A. Nawaz, S. Shujah, Appl. Organomet. Chem. 37 (2023) e7161 (https://doi.org/10.1002/aoc.7161)
S. Saroya, S. Asija, N. Kumar, Y. Deswal, J. Indian Chem. Soc. 99 (2022) 100379 (https://doi.org/10.1016/j.jics.2022.100379)
A. Sharma, A. Jain, S. Saxena, Appl. Organomet. Chem. 29 (2015) 499 (https://doi.org/10.1002/aoc.3321)
A. Boora, J. Devi, B. Kumar, B. Taxak, BioMetals 38 (2025) 153 (https://doi.org/10.1007/s10534-024-00644-8)
A. Kumawat, K. Soni, S. Saxena, A. Jain, Appl. Organomet. Chem. C 37 (2023) e7205 (https://doi.org/10.1002/aoc.7205)
P. Debnath, K. S. Singh, T. S. Devi, S. S. Singh, R. J. Butcher, L. Sieroń, W. Maniukiewicz, Inorg. Chim. Acta 510 (2020) 119736 (https://doi.org/10.1016/j.ica.2020.119736)
K. Soni, A. Kumawat, S. Saxena, A. Jain, Bull. Chem. Soc. Jpn. 97 (2024) 9 (https://doi.org/10.1093/bulcsj/uoae056)
A. M. Kumar, S. Budania, A. Jain, Synth. Commun. 54 (2024) 390 (https://doi.org/10.1080/00397911.2024.2306641)
S. Saroya, S. Asija, N. Kumar, Y. Deswal, J. Indian Chem. Soc. 99 (2022) 100379 (https://doi.org/10.1016/j.jics.2022.100379)
W. T. Piver, Environ. Health Persp. 4 (1973) 61 (https://ehp.niehs.nih.gov/doi/pdf/10.1289/ehp.730461)
M. Hoch, Appl. Geochem. 16 (2001) 719 (https://doi.org/10.1016/S0883-2927(00)00067-6)
S. Iftikhar, S. Hussain, S. Murtaza, D. Ali, S. Yousuf, M. A. Ali, A. Haider, M. Shahid, A. M. Alsuhaibani, M. S. Refat, Appl. Organomet. Chem. 38 (2024) e7581 (https://doi.org/10.1002/aoc.7581)
Y. Tan, Z. Zhang, J. Liu, Y. Tan, W. Jiang, J. Mol. Struct. 1322 (2025) 140697 (https://doi.org/10.1016/j.molstruc.2024.140697)
L. Pellerito, L. Nagy, Coord. Chem. Rev. 224 (2002) 111 (https://doi.org/10.1016/S0010-8545(01)00399-X)
F. Marchetti, C. Pettinari, C. Di Nicola, A. Tombesi, R. Pettinari, Coord. Chem. Rev. 401 (2019) 213069 (https://doi.org/10.1016/j.ccr.2019.213069)
M. Sirajuddin, S. Ali, A. Shahnawaz, F. Perveen, S. Andleeb, S. Ali, J. Mol. Struct. 1207 (2020) 127809 (https://doi.org/10.1016/j.molstruc.2020.127809)
B. S. Jensen, Acta Chem. Scand. 13 (1959) 1668 (https://doi.org/10.3891/acta.chem.scand.13-1668)
S. Sharma, A. Jain, S. Saxena, Main Group Met. Chem. 30 (2007) 63 (https://doi.org/10.1515/MGMC.2007.30.2-3.63)
G. B. Deacon, R. J. Phillips, Coord. Chem. Rev. 33 (1980) 227 (https://doi.org/10.1016/S0010-8545(00)80455-5)
R. K. Gupta, A. Jain, S. Saxena, Main Group Met. Chem. 33 (2010) 167 (https://doi.org/10.1515/mgmc.2010.33.4-5.167)
A. Joshi, S. Verma, A. Jain, S. Saxena, Main Group Met. Chem. 28 (2005) 31 (https://doi.org/10.1515/MGMC.2005.28.1.31)
S. Budania, S. Saxena, A. Jain, IOP Conf. Ser. Mater. Sci. Eng. 1248 (2022) 012106 (https://doi.org/10.1088/1757-899X/1248/1/012106).