Syntheses and computational analyses of selected macrolide derivatives derived from clarithromycin A
Main Article Content
Abstract
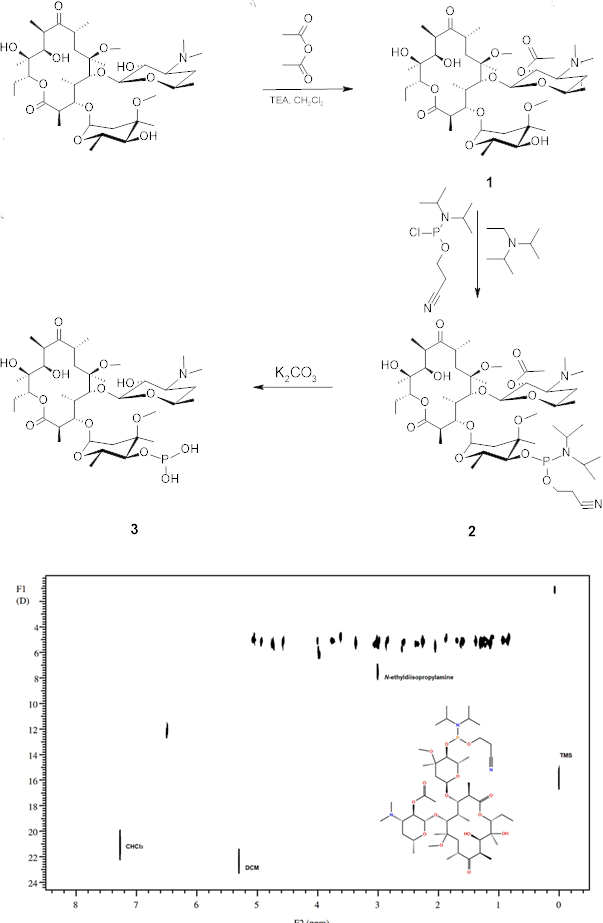
This study presents the synthesis and experimental and computational analysis of novel macrolide derivatives obtained from clarithromycin A, with the aim of exploring their potential to address the growing problem of antimicrobial resistance. The compounds synthesized include 2′-O-acetyl-clarithromycin A, its phosphoramidite derivative, and the corresponding phosphonyl derivative. Special attention was paid to the optimization of phosphitylation conditions due to the inherent instability of phosphoramidite compounds. The purity of the phosphoramidite derivative was successfully confirmed using diffusion-ordered NMR spectroscopy (DOSY). Comprehensive conformational analyses were carried out using molecular modeling techniques, followed by molecular docking and MM-GBSA calculations with a target protein from Escherichia coli to evaluate the relative binding affinities of clarithromycin A and its derivatives. The results indicate that the phosphoramidite and phosphonyl derivatives exhibit comparable binding affinities relative to the parent antibiotic. In addition, complex mass spectrometric fragmentation patterns of the phosphorus-containing derivatives were analyzed and rationalized using the MS Fragmenter computational tool.
Downloads
Metrics
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.

Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution license 4.0 that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
Funding data
-
Ministarstvo Prosvete, Nauke i Tehnološkog Razvoja
Grant numbers 451-03-47/2023-01/200124;451-03-66/2024-03/200124;451-03-136/2025-03/200124
References
Antimicrobial resistance, https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance, accessed on the 19th February 2026
I. Kyriakidis, E. Vasileiou, Z. D. Pana, A. Tragiannidis, Pathogens 10 (2021) 373 (https://doi.org/10.3390/pathogens10030373)
A. K. Talley, A. Thurston, G. Moore, V. K. Gupta, M. Satterfield, E. Manyak, S. Stokes, A. Dane, D. Melnick, Antimicrob. Agents Chemother. 65 (2021) (https://doi.org/10.1128/aac.01208-21)
T. P. Prakash, A. M. Kawasaki, A. S. Fraser, G. Vasquez, M. Manoharan, J. Org. Chem. 67 (2002) 357 (https://doi.org/10.1021/jo0103975)
C. Xie, M. A. Staszak, J. T. Quatroche, C. D. Sturgill, V. V. Khau, M. J. Martinelli, Org. Process Res. Dev. 9 (2005) 730 (https://doi.org/10.1021/op050077d)
C. Wagner, H.-A. Wagenknecht, Chem. Eur. J. 11 (2005) 1871 (https://doi.org/10.1002/chem.200401013)
G. Hofle, W. Steglich, H. Vorbriiggen, Angew. Chem. Int. Ed. Engl. 17 (1978) 569 (https://doi.org/10.1002/anie.197805691)
A. P. Guzaev, M. Manoharan, J. Org. Chem. 66 (2001) 1798 (https://doi.org/10.1021/jo001591e)
W. H. Kuijpers, J. Huskens, L. H. Koole, C. A. van Boeckel, Nucleic Acids Res. 18 (1990) 5197 (https://doi.org/10.1093/nar/18.17.5197)
N. D. Sinha, J. Biernat, H. Köster, Tetrahedron Lett. 24(52) (1983) 5843 (https://doi.org/10.1016/S0040-4039(00)94216-3)
V. Gilard, S. Trefi, S. Balayssac, M.-A. Delsuc, T. Gostan, M. Malet-Martino, R. Martino, Y. Prigent, F. Taulelle, Chapter 6 - DOSY NMR for drug analysis, in NMR Spectroscopy in Pharmaceutical Analysis, U. Holzgrabe, I. Wawer, B. Diehl, Eds., Elsevier Science, 2008, p. 269-289 (https://doi.org/10.1016/B978-0-444-53173-5.00011-1)
H. Barjat, G. A. Morris, S. Smart, A. G. Swanson, S. C. R. William, J. Mag. Res. Ser. A 116 (1995) 206 (https://doi.org/10.1006/jmra.1995.0009)
H. Barjat, G. A. Morris, S. Smart, A. G. Swanson, S. C. R. William, J. Mag. Res. Ser. B 108 (1995) 170 (https://doi.org/10.1006/jmrb.1995.1118)
G. A. Morris, Diffusion-Ordered Spectroscopy (DOSY). Encyclopedia of Nuclear Magnetic Resonance: Advances in NMR, John Wiley and Sons, 2009 (https://doi.org/10.1002/9780470034590.emrstm0119.pub2)
W. R. Baker, J. D. Clark, R. L. Stephens, K. H. Kim, J. Org. Chem. 53 (1988) 2340 (https://doi.org/10.1021/jo00245a038)
W. D. Cornell, P. Cieplak, C. I. Bayly, I. R. Gould, K. M. Merz, Jr., D. M. Ferguson, D. C. Spellmeyer, T. Fox, J. W. Caldwell, P. A. Kollman, J. Am. Chem. Soc. 117 (1995) 5179 (https://doi.org/10.1021/ja00124a002)
B. Arsic, A. Awan, R. J. Brennan, J. A. Aguilar, R. Ledder, A. J. McBain, A. C. Regan, J. Barber, MedChemCommun. 5 (2014) 1347 (https://doi.org/10.1039/c4md00220b)
K. S. Watts, P. Dalal, A. J. Tebben, D. L. Cheney, J. C. Shelley, J. Chem. Inf. Model. 54(10) (2014) 2680 (https://doi.org/10.1021/ci5001696)
J. A. Dunkle, L. Xiong, A.S. Mankin, J. H. D. Cate, Proc. Natl. Acad. Sci. U.S.A. 107(40) (2010) 17152 (https://doi.org/10.1073/pnas.1007988107)
MS Fragmenter, version 2023.1.1, Advanced Chemistry Development, Inc. (ACD/Labs), Toronto, ON, Canada, www.acdlabs.com.
Y. Qin, M. Sun, N. Zhang, Y. Yang, P. Ma, Bioorg. Chem. 127 (2022) 106020 (https://doi.org/10.1016/j.bioorg.2022.106020)
A. F. Sandahl, T. J. D. Nguyen, R. A. Hansen, M. B. Johansen, T. Skrydstrup, K. V. Gothelf, Nat. Commun. 12 (2021) 2760 (https://doi.org/10.1038/s41467-021-22945-z)
A. Ambrus, D. Yang, Anal. Biochem. 367 (2007) 56 (https://doi.org/10.1016/j.ab.2007.04.025)
E. J. Cabrita, S. Berger, Magn. Reson. Chem. 40 (2002) S122 (https://doi.org/10.1002/mrc.1082)
E. J. Cabrita, S. Berger, P. Brauer, J. Karger, J. Magn. Reson. 157 (2002) 124 (https://doi.org/10.1006/jmre.2002.2574)
M. N. Mordi, M. D. Pelta, V. Boote, G. A. Morris, J. Barber, J. Med. Chem. 43 (2000) 467 (https://doi.org/10.1021/jm9904811)
J. R. Everett, J. W. Tyler, J. Chem. Soc. Perkin Trans. 2 11 (1987) 1659 (https://doi.org/10.1039/P29870001659)
J. Poehlsgaard, S. Douthwaite, Nat Rev Microbiol. 3 (2005) 870 (https://doi.org/10.1038/nrmicro1265)
E. Wang, H. Sun, J. Wang, Z. Wang, H. Liu, J. Z. H. Zhang, T. Hou, Chem. Rev. 119 (2019) 9478 (https://doi.org/10.1021/acs.chemrev.9b00055)
G. Poli, C. Granchi, F. Rizzolio, T. Tuccinardi, Molecules 25(8) (2020) 1971 (https://doi.org/10.3390/molecules25081971)
G. Rastelli, L. Pinzi, Front. Chem. 7 (2019) 498 (https://doi.org/10.3389/fchem.2019.00498).