Epoxy- and cyclopropane-functional copolymers: synthesis, thermal properties, and photocrosslinking behavior
Main Article Content
Abstract
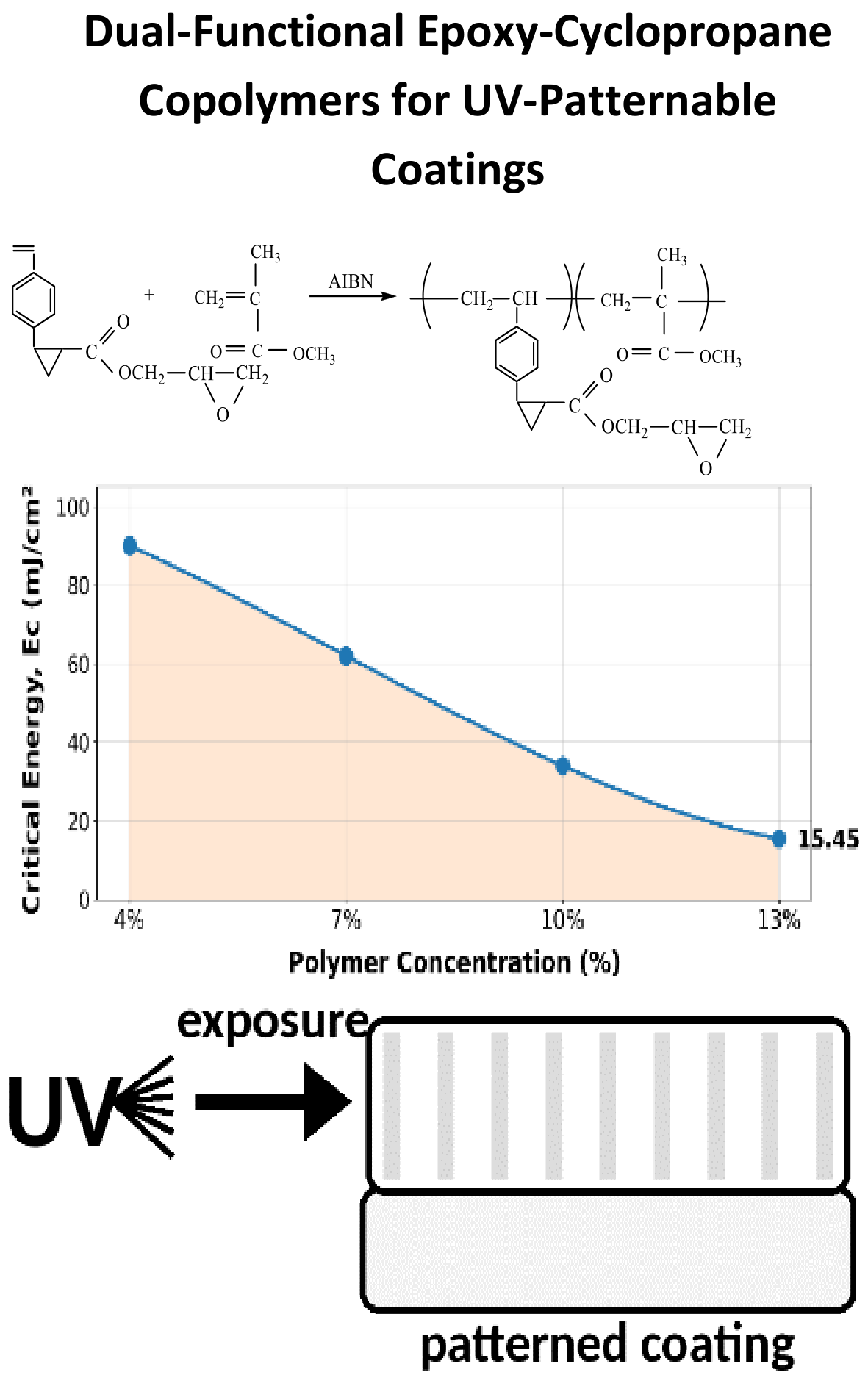
Copolymers bearing both epoxy and cyclopropane groups were synthesized by free-radical copolymerization of glycidyl 2-(4-vinylphenyl)cyclopropanecarboxylate (GVPCC) with methyl methacrylate (MMA) using AIBN at 343 K, in bulk and in benzene under inert atmosphere. Copolymer compositions were determined by spectroscopy, and copolymerization parameters were evaluated by the Fineman–Ross method. The reactivity ratios were r1(GVPCC) = 0.68 ± 0.05 and r2(MMA) = 0.51 ± 0.07; their product (r1∙r2 = 0.35) indicates random copolymerization with a tendency toward alternation. Alfrey–Price parameters (Q1 = 0.96, e1 = −0.63; Q2 = 0.74, e2 = 0.40) confirm strong comonomer interactions and pronounced polar effects. For a 50/50 copolymer, the intrinsic viscosity was 0.66 dL g-1 (benzene, 25 °C). Thermogravimetric analysis showed composition-dependent stability with T5 = 250–320 °C, increasing with GVPCC content, alongside improved adhesion (up to 5.6 MPa) and Vicat softening temperature (121 °C). UV irradiation produced efficient crosslinking and negative-tone photoresist behavior (resolution with depth of penetration, Dp = 0.25–0.35 μm; critical exposure energy, Ec = 14.5–16.4 mJ cm-2; sensitivity, S = 61–69 cm2 J-1), demonstrating potential for UV-patternable microfabrication materials.
Downloads
Metrics
Article Details

This work is licensed under a Creative Commons Attribution 4.0 International License.

Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution license 4.0 that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal.
References
Q. Lin, Polymer 286 (2023) 126395 (https://doi.org/10.1016/j.polymer.2023.126395)
C. Ober, K. Käfer, F. Yuan, Ch., Polymer 280 (2023) 126020 (https://doi.org/10.1016/j.polymer.2023.126020)
R. Zhou, M. Cao, Y. Tan, M. Neisser, H. Xu, Sci. Adv. 11(29) (2025) 1918 (https://doi.org/10.1126/sciadv.adx1918)
Y. Wang, H. Yu, L. Wang, Y. Zhang, Z. Zhu, Y. Zhang, Y. Lu, Ch. Ouyang, J. Mat. Chem. A 13(36) (2025) 29860-29884 (https://doi.org/10.1039/D5TA04194E)
Y. J. Wan, G. Li, Y. M. Yao, X. L. Zeng, P. L. Zhu, R. Sun, Comp. Comm. 19 (2020) 154-167 (https://doi.org/10.1016/j.coco.2020.03.011)
Y. Wen, C. Chen, Y. Ye, Z. Xue, H. Liu, X. Zhou, Y. Zhang, D. Li, X. Xie, Y. Mai, Adv. Mat. 34(52) (2022) e2201023. (https://doi.org/10.1002/adma.202201023)
K. G. Guliyev, G. Z. Ponomareva, Kh. G. Nazaraliyev, A. M. Guliyev, Azerb. Khimicheskiy Zhurnal 1 (1999) 87 (in Russian)
K. G. Guliev, G. Z. Ponomareva, Kh. G. Nazaraliev, A. M. Guliev, Azerb. Khimicheskiy Zhurnal 1 (1999) 87–90 (in Russian)
K.G. Guliyev, S.B. Mamedli, T.S.D. Gulverdashvili, A.M. Guliyev, Book: Applied Chemistry and Chemical Engineering, İmprint: Apple Academic Press., Volume 2, 2017
A. I. Sadygova, Azerbaijan Chem. J. 3 (2022) 45–50 (https://akj.az/en/journals/949)
M. Sayes, G. Benoit, A. B. Charette, Ang. Chem. Int. Ed. 57 (2018) 13514-13518 (https://doi.org/10.1002/anie.201807347)
K. Mizuno, N. Ichinose, Y. Yoshimi, J. Photochem. Photobiol. C: Photochem. Rev. 1(2) (2000) 167-193 (https://doi.org/10.1016/S1389-5567(00)00011-3)
K.G. Guliev, G.Z. Ponomareva, A. M. Guliev, Vysokomolekulyarnye soedineniya, Seriya B 49(8) (2007) 1577–1581(in Russian)
M. Fineman, S. D. Ross, J. Polymer Sci. 5 (1950) 259–262 (https://doi.org/10.1002/pol.1950.120050210)
F. R. Mayo, F. M. Lewis, J. Am. Chem. Soc. 66 (1944) 1594–1601 (https://doi.org/10.1021/ja01237a052)
T. Alfrey Jr., C. C. Price, J. Polymer Sci. 2 (1947) 101–106 (https://doi.org/10.1002/pol.1947.120020112)
Y. Zhang, Y. Li, W. Liu, Adv. Funct. Mat. 25(3) (2015) 471-480 (https://doi.org/10.1002/adfm.201401989)
J. Bennett, Addit. Manuf. 18 (2017) 203–212 (https://doi.org/10.1016/j.addma.2017.10.009).